Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- Structural biology is solved -- now what?
- XFEL Pulses Demonstrate How Plants Perceive Light
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
- Details
- Friday, 08 September 2023

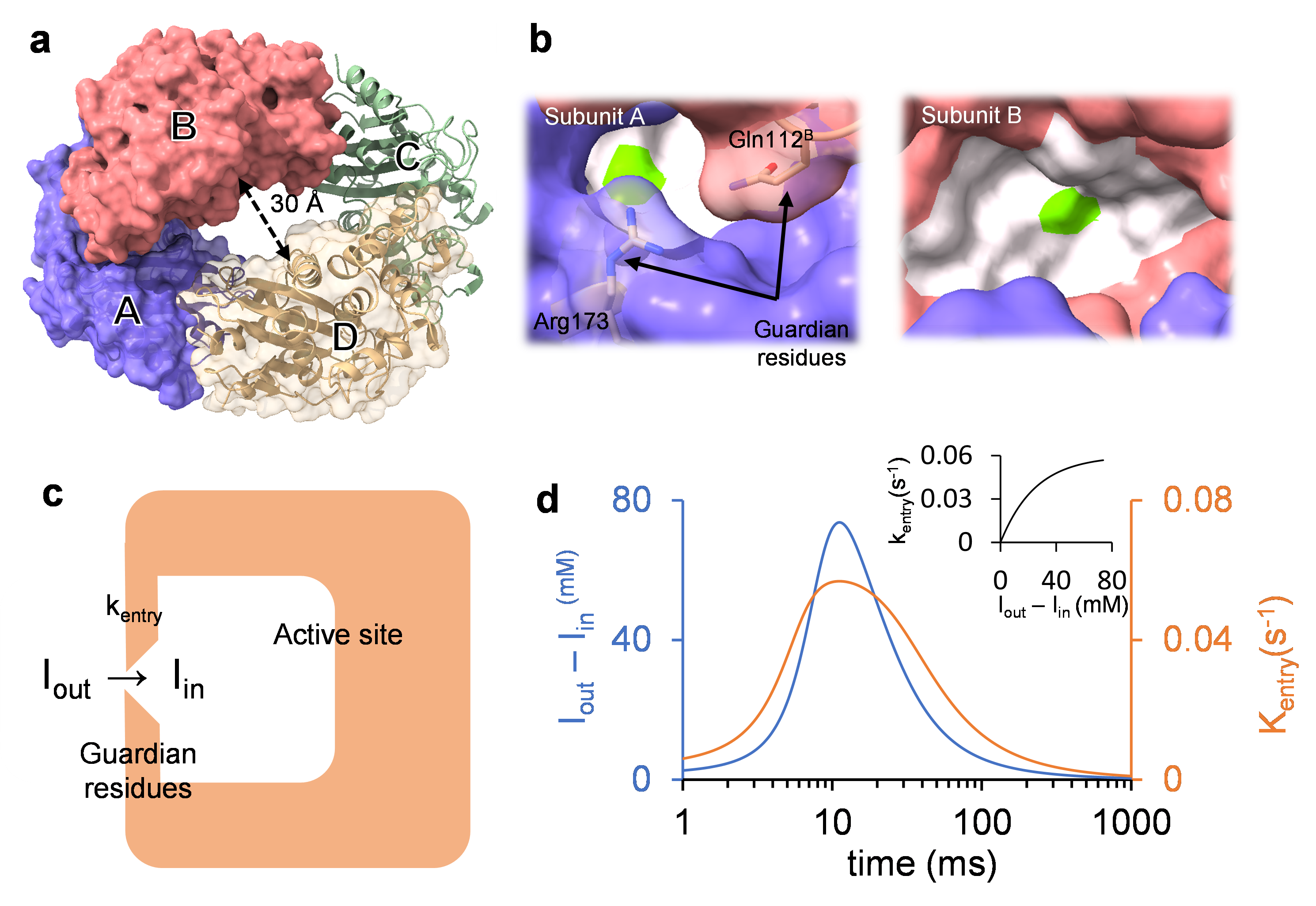
In a paper just published in Nature Communications, a team of BioXFEL researchers and collaborators used mix-and-inject serial crystallography (MISC) to analyze the reaction of the β-lactamase BlaC from tuberculosis bacteria with the suicide inhibitor sulbactam (SUB).
In this study, Tek Malla, a graduate student from Marius Schmidt‘s lab at the University of Wisconsin Milwaukee, mixed microcrystals of BlaC with SUB and injected the mixture after a delay into intense X-ray pulses from the X-ray Free Electron Laser LCLS. Injectors were provided by Kara Zielinksi from Lois Pollack’s lab at Cornell University. The experiment was performed at the MFX instrument at LCLS. The research is insofar remarkable since the experiment at LCLS has been performed in the middle of the COVID-19 pandemic. Schmidt, Pollack and other collaborators kept their labs open during these difficult times. Malla most bravely flew to California and was the only external user for this particular beamtime. The experiment would not have been possible without strong support by the LCLS staff who kept the XFEL operational and supported the experiment as much as possible, and by the Stanford Linear Accelerator Center leadership who implemented safety measures to protect all parties.
The outcome of the experiment is remarkable. An entire time-series of MISC data was collected from 3 ms to 700 ms. The time-series of X-ray data was analyzed by an algorithm with the singular value decomposition (SVD) at its base. This algorithm continues to work when unit cell changes make the calculation of isomorphous difference maps impossible. Using the SVD analysis, kinetic evidence for ligand binding heterogeneity, ligand gating, cooperativity, induced fit, and conformational selection was obtained all from the same set of MISC data, detailing how SUB approaches the catalytic clefts and binds to the enzyme noncovalently before reacting to a trans-enamine.
The bio-medical significance of the reaction catalyzed by BlaC highlights the importance of this research and the meaningfulness of the mix-and-inject approach. The publication [1] is open access and can be retrieved from https://doi.org/10.1038/s41467-023-41246-1.
[1] T.N. Malla, K. Zielinski, L. Aldama, S. Bajt, D. Feliz, B. Hayes, M. Hunter, C. Kupitz, S. Lisova, J. Knoska, J. M. Martin-Garcia, V. Mariani, S. Pandey, I. Poudyal, R. G. Sierra, A. Tolstikova, O. Yefanov, C.-H. Yoon, A. Ourmazd, P. Fromme, P. Schwander, A. Barty, H. N. Chapman, E. A. Stojkovic, A. Batyuk, S. Boutet, G. N. Phillips, Jr., L. Pollack, M. Schmidt (2023) Heterogeneity in the M. tuberculosis β-Lactamase Inhibition by Sulbactam, Nature Communications, published online on Sep 7 2023, 10.1038/s41467-023-41246-1.





