Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- Structural biology is solved -- now what?
- XFEL Pulses Demonstrate How Plants Perceive Light
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
- Details
- Friday, 06 May 2016
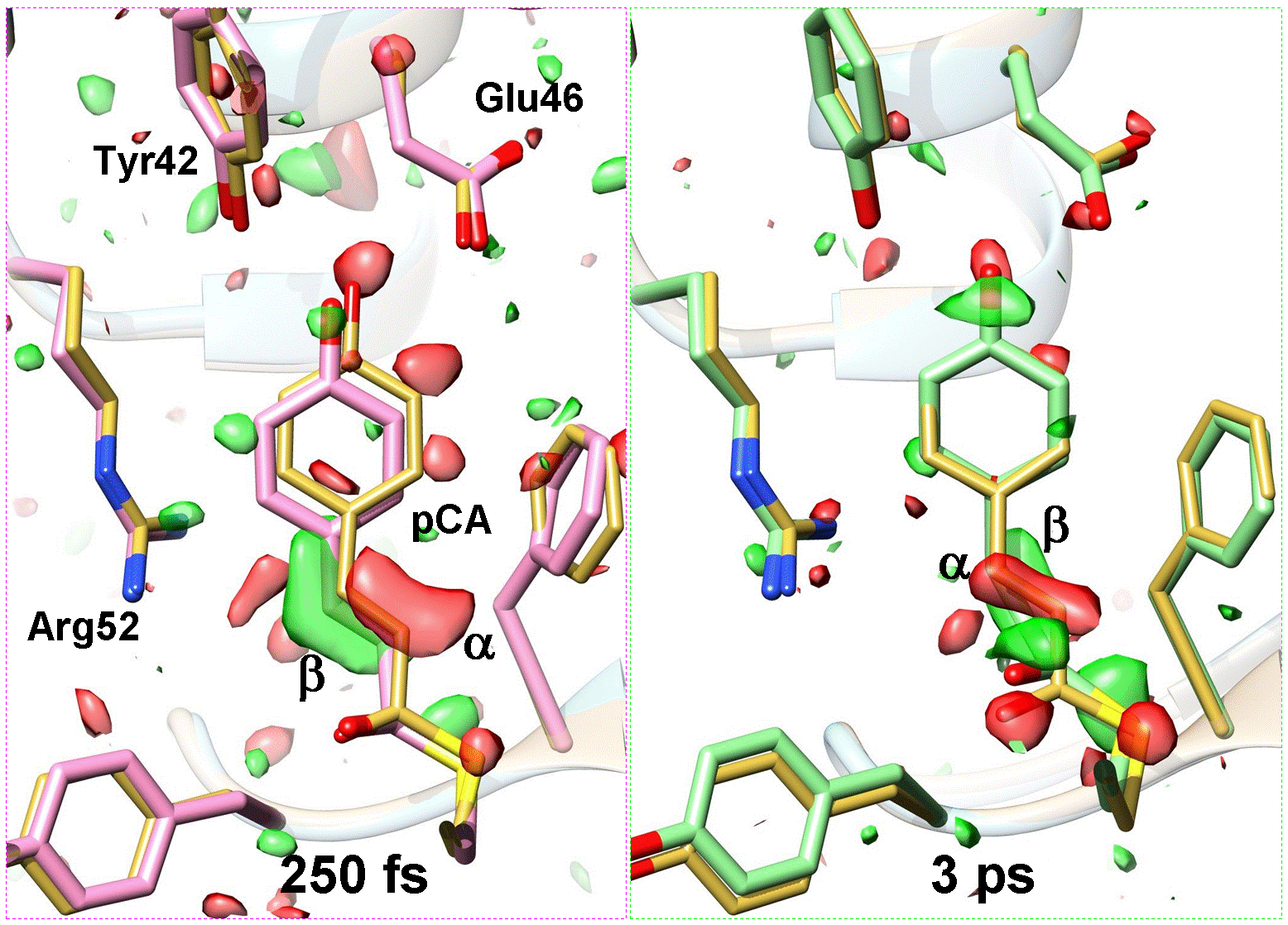
Researchers for years have been attempting to look at ultrafast motions at the atomic level, for example how light gets transferred into energy by plants during photosynthesis. Finally, with the use of an X-ray Free Electron Laser at Stanford, NSF BioXFEL Science and Technology researchers and collaborators from three other universities have been successful in fully exploiting the power of this instrumentation and have created a molecular motion ‘movie’ on the femtosecond time scale.
A femtosecond is a thousandth of a millionth of a millionth of a second. The high power X-ray laser allows for some of the fastest ‘snapshots’ that have ever been taken. Ultimately, this group lead by Marius Schmidt, a physicist at the University of Wisconsin, Milwaukee and a partner in the NSF BioXFEL Science and Technology Center, has identified the fundamental motions of how light effects a light sensitive biomolecule, and how it triggers the structural changes necessary for signal transduction.
The results build on work from just over a year ago, also led by Schmidt, which was the first to demonstrate the capabilities of this instrumentation and the power of interdisciplinary collaboration. The current study also shows the ability to capture motion at the atomic level, but on scales about 100,000 times faster than the previous study. This work opens the doors for many biological targets to be investigated this way. “With an exposure time of about 200 femtoseconds, this is surely the fastest camera in the world. And the process we capture is closely similar to the first event in human vision, when a light photon strikes the back of your eye. We see the molecules moving in response to the arrival of that quantum of light,” said BioXFEL STC Science Director, John Spence, a physics professor at Arizona State University.
“The absorption of light leaves PYP in an excited state from which it relaxes very quickly. It does so by rearranging its atomic structure in what is known as a trans-to-cis isomerisation. We’re the first to succeed in taking real-time snapshots of the fundamental processes of this type of reaction. A very similar reaction also occurs in the retina of our eye and enables us to see,” said Schmidt.
“This research, which was born from a group of scientist who are collaborating as part of the National Science Foundation Science and Technology Centers program represent the best in collaborative, interdisciplinary research,” said Dragana Brzakovic, of the Office of Integrative Activities at NSF, “The NSF established this program in 1987 to support innovative, break-through research. This Center, as well as the other 13 currently operating across the country bring together top talent to solve complex problems. This is the perfect example of one of their many contributions.”
The full paper can be found in Science- Femtosecond structural dynamics drives the trans/cis isomerization in photoactive yellow protein





