Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- Structural biology is solved -- now what?
- XFEL Pulses Demonstrate How Plants Perceive Light
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
- Details
- Tuesday, 11 April 2017

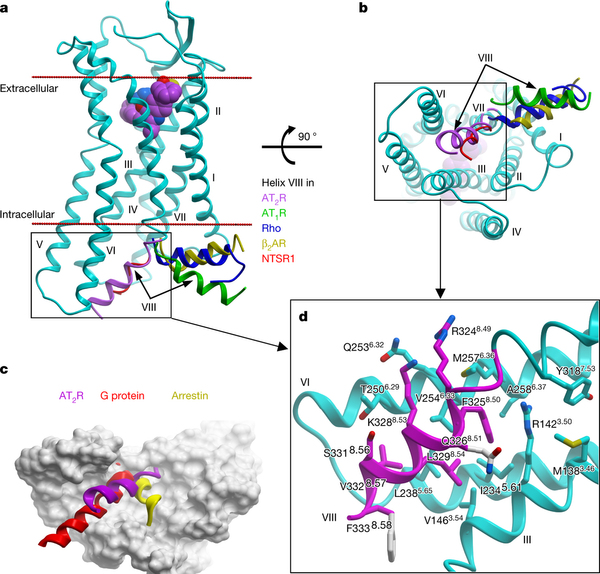
Designing molecules that selectively bind to a specific receptor type is often challenging, but can be crucial for different therapeutic purposes. Both angiotensin II receptors are important drug targets, since the blockade of AT1R has anti-hypertensive effects, while the modulation of AT2R could be useful for cardioprotection, neuropathic pain relief and the treatment of several other conditions.
Although the AT1R and AT2R ligands share common scaffolds, the ligand-binding pockets of these two receptors are markedly different, and these differences could be exploited for designing selective ligands. The AT2R crystal structures determined in this study improve our understanding of the two types of the human angiotensin receptor and provide new insights into the structural basis for the binding and selectivity of small molecules of therapeutic significance. Our results are therefore expected to facilitate the rational structure-based drug design for improved selectivity.





