Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- Structural biology is solved -- now what?
- XFEL Pulses Demonstrate How Plants Perceive Light
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
- Details
- Tuesday, 30 May 2017

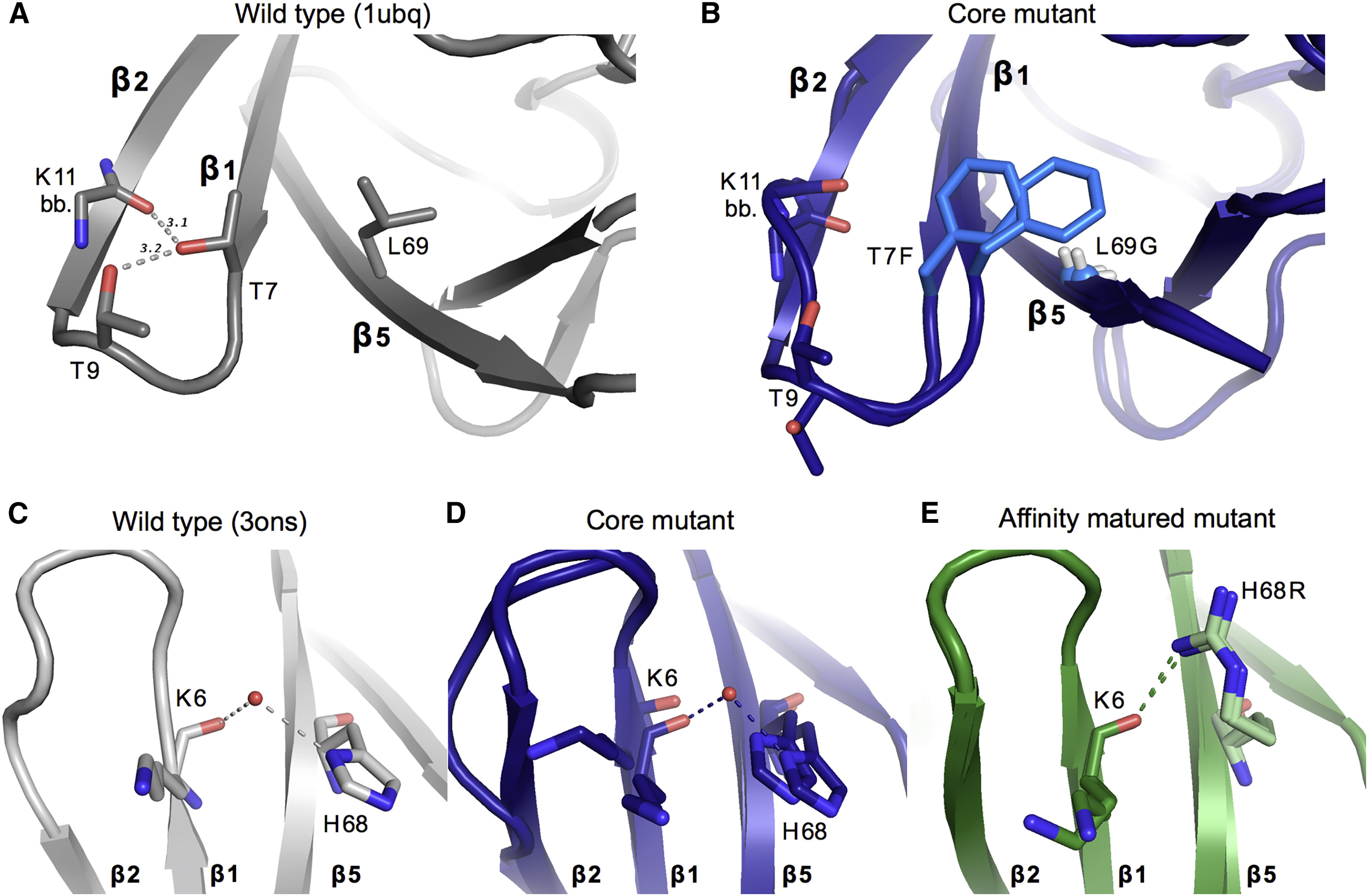
Although protein design has been used to introduce new functions, designed variants generally only function as well as natural proteins after rounds of laboratory evolution. One possibility for this pattern is that designed mutants frequently sample nonfunctional conformations.
To test this idea, we exploited advances in multiconformer modeling of room-temperature X-ray data collection on redesigned ubiquitin variants selected for increasing binding affinity to the deubiquitinase USP7. Initial core mutations disrupt natural packing and lead to increased flexibility. Additional, experimentally selected mutations quenched conformational heterogeneity through new stabilizing interactions. Stabilizing interactions, such as cation-pi stacking and ordered waters, which are not included in standard protein design energy functions, can create specific interactions that have long-range effects on flexibility across the protein. Our results suggest that increasing flexibility may be a useful strategy to escape local minima during initial directed evolution and protein design steps when creating new functions.





