Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- XFEL Pulses Demonstrate How Plants Perceive Light
- Structural biology is solved -- now what?
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
- Details
- Thursday, 03 August 2017

BioXFEL scientist Marc Messerschmidt, along with his colleagues, published a research article in OSA publishing.
Summary:
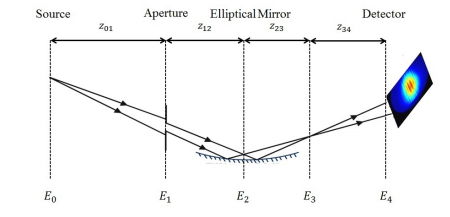
Determining fluctuations in focus properties is essential for many experiments at Self-Amplified-Spontaneous-Emission (SASE) based Free-Electron-Lasers (FELs), in particular for imaging single non-crystalline biological particles.
We report on a diffractive imaging technique to fully characterize highly focused, single-shot pulses using an iterative phase retrieval algorithm, and benchmark it against an existing Hartmann wavefront sensor. The results, both theoretical and experimental, demonstrate the effectiveness of this technique to provide a comprehensive and convenient shot-to-shot measurement of focused-pulse wave fields and source-point positional variations without the need for manipulative optics between the focus and the detector.
The advent of hard X-ray Free-Electron-lasers (XFEL), such as the European XFEL in the Hamburg area, Germany, the Linac Coherent Light Source (LCLS), CA, USA or the SPring-8 Angstrom Compact free electron LAser (SACLA), Japan, enables a broad range of novel experiments including single-shot diffraction imaging of biological structures and time-resolved imaging of the dynamics of matter





