Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- XFEL Pulses Demonstrate How Plants Perceive Light
- Structural biology is solved -- now what?
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
News
- Details
- Wednesday, 03 October 2018
An international collaboration led by the Deutsches Elektronen-Synchrotron or DESY, with participation from Arizona State University's Biodesign Center for Applied Structural Discovery, the Department of Physics and the School of Molecular Sciences, as well as the University of Wisconsin Milwaukee Physics Department has announced the results of the first scientific experiments at Europe's new X-ray free-electron laser, European XFEL.
- Details
- Wednesday, 03 October 2018

The contest — founded by a UB chemist — reaches thousands of children around the country each year
- Details
- Wednesday, 26 September 2018

A research consortium led by the University at Buffalo has been awarded $22.5 million from the National Science Foundation (NSF) to continue its groundbreaking work developing advanced imaging techniques for critical biological processes that are difficult, if not impossible, to see with conventional methods.
- Details
- Tuesday, 25 September 2018

Structure Biologist (f/m/d) in the field of X-ray Crystallography
- Details
- Tuesday, 25 September 2018

Srd Div. Director [Senior/ Distinguished Staff Scientist]
- Details
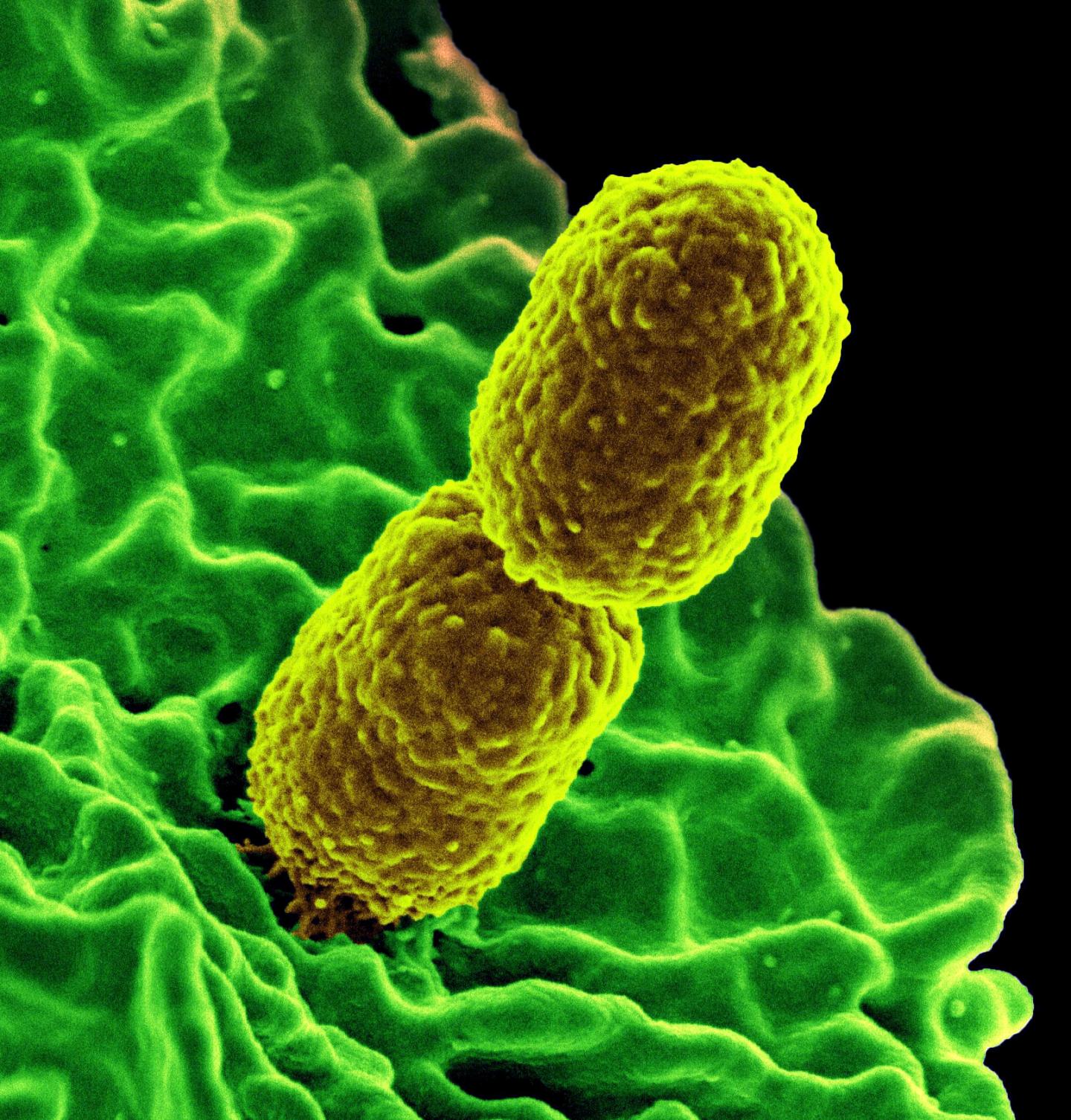
- Monday, 10 September 2018
Myxobacteria are swarming micro-organisms that, under starving conditions, form multicellular aggregates called fruiting bodies. The fruiting body formation is, in addition, light dependent.





