Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- Structural biology is solved -- now what?
- XFEL Pulses Demonstrate How Plants Perceive Light
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
News
- Details
- Tuesday, 01 August 2017
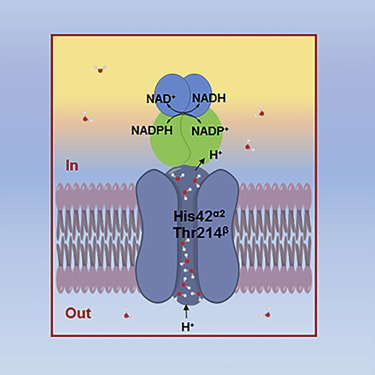
BioXFEL scientist Vadim Cherezov, along with others, publishes research article in ScienceDirect.
Abstract:
The nicotinamide nucleotide transhydrogenase (TH) is an integral membrane enzyme that uses the proton-motive force to drive hydride transfer from NADH to NADP+ in bacteria and eukaryotes.
- Details
- Thursday, 27 July 2017
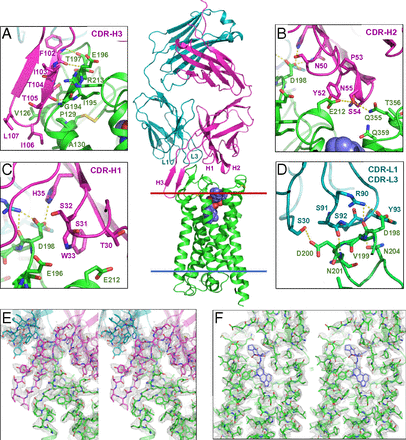
Monoclonal antibodies provide an attractive alternative to small-molecule therapies for a wide range of diseases. Given the importance of G protein-coupled receptors (GPCRs) as pharmaceutical targets, there has been an immense interest in developing therapeutic monoclonal antibodies that act on GPCRs.
- Details
- Tuesday, 25 July 2017
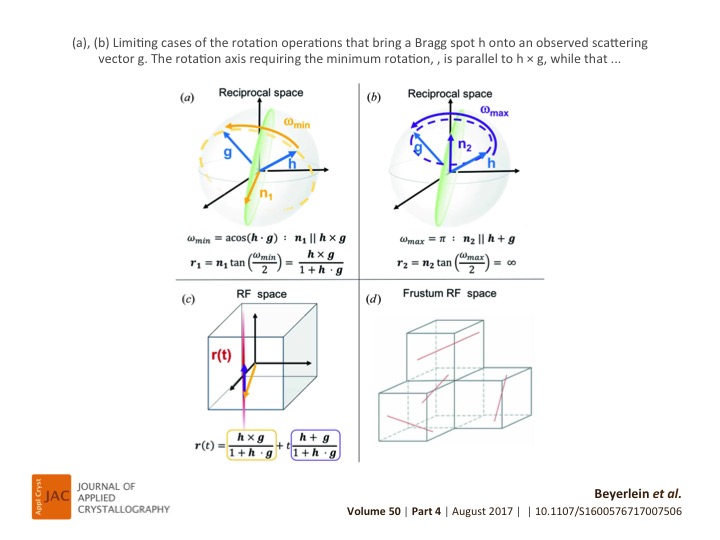
A novel algorithm for indexing multiple crystals in snapshot X-ray diffraction images, especially suited for serial crystallography data, is presented. The algorithm, FELIX, utilizes a generalized parametrization of the Rodrigues–Frank space, in which all crystal systems can be represented without singularities.
- Details
- Tuesday, 18 July 2017
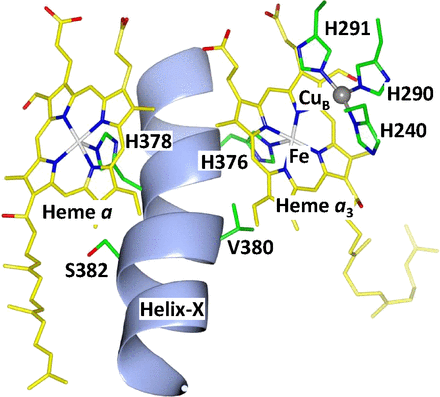
Cytochrome c oxidase (CcO), the terminal enzyme in the electron transfer chain, translocates protons across the inner mitochondrial membrane by harnessing the free energy generated by the reduction of oxygen to water.
- Details
- Wednesday, 12 July 2017
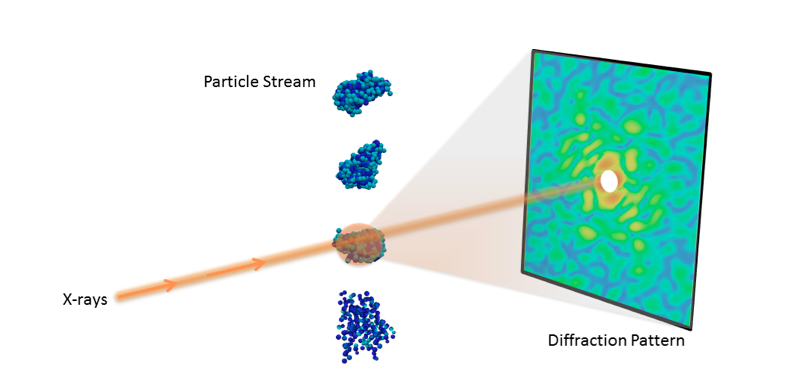
Understanding the 3D molecular structure of important nano-objects like proteins and viruses is crucial in biology and medicine. With recent advances in X-ray technology, scientists can now collect diffraction images from individual particles, ultimately allowing researchers to visualize molecules at room temperature.
- Details
- Tuesday, 11 July 2017
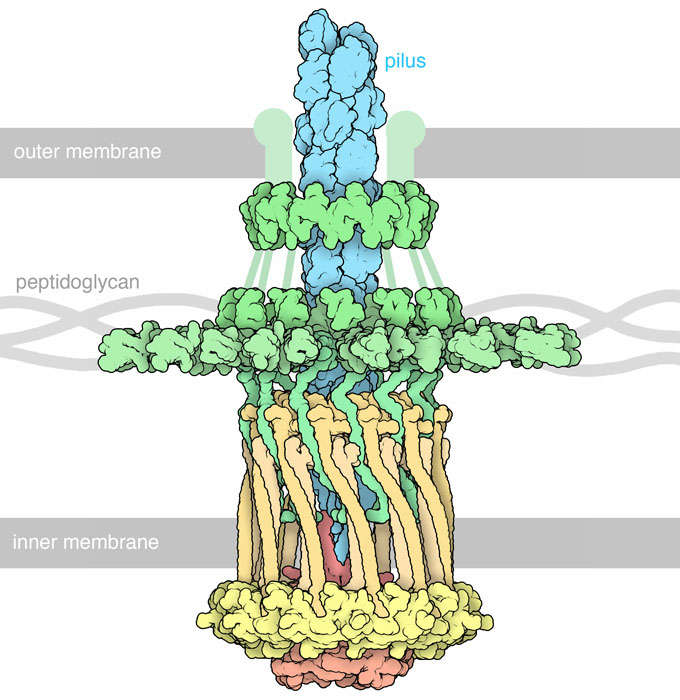
Bacteria live in complex ecologies, filled with competitors, environmental dangers, and defensive hosts. They face these challenges in many ways: by gathering together into tough biofilm communities, by sharing genetic information encoding useful proteins, and by picking up and moving when things look better elsewhere.
More Articles...
- SLAC's Electron Hub Gets New 'Metro Map' for World's Most Powerful X-Ray Laser
- European XFEL enters operation phase
- Adenine Riboswitch in Action
- X-ray laser diffraction for structure determination of the rhodopsin-arrestin complex
- Preparation of microcrystals in lipidic cubic phase for serial femtosecond crystallography





