Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- XFEL Pulses Demonstrate How Plants Perceive Light
- Structural biology is solved -- now what?
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
News
- Details
- Tuesday, 20 February 2018

DOE funded postdoc position for the study of photosynthesis proteins and enzymes using terahertz light. We are seeking a highly independent and skilled researcher to join our effort to establish THz techniques that characterize protein structural dynamics.
- Details
- Monday, 29 January 2018
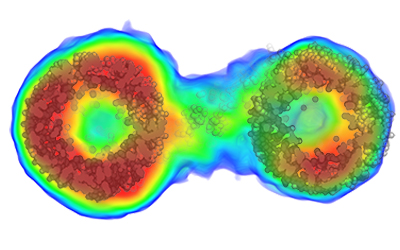
New research published today in Nature Methods by BioXFEL researcher Tom Grant opens up new avenues for studying the structures of particles floating in solution.
Typically, molecular structures are determined using a technique known as crystallography, where molecules are chemically induced to align next to each other in a large 3D lattice.
- Details
- Thursday, 02 November 2017

BioXFEL was well represented at the LCLS/SSRL Users' Meeting with Henry Chapman giving a plenary talk on Serial Femtosecond Crystallography: Past, Present, and Future featuring snippets from data as recent as 24 hours before the talk. David Bushnell, moderated a session that included the LCLS young investigator award and Linda Horton, Director of the Department of Energy Basic Energy Sciences Materials Science and Engineering Division. A meeting of the joint LCLS and SSRL Users Executive Committee took place with Linda Horton, Petra Fromme, David Bushnell and Edward Snell present.
- Details
- Tuesday, 31 October 2017

A team of BioXFEL ASU scientists led by Professor and STC Member Alexandra Ros in the School of Molecular Sciences and the Biodesign Center for Applied Structural Discovery, has been just the second user group to conduct experiments at the brand new European X-ray free electron laser facility (EuXFEL) in Hamburg, Germany. This 1.5-billion-dollar facility is the third, and by far the most powerful, X-ray laser in the world. After ten years of construction, it opened for first experiments just a month ago.
- Details
- Thursday, 05 October 2017

On October 4th, 2017, Drs. Jacques Dubochet (formerly of University of Lausanne in Switzerland), Joachim Frank (Columbia University), and Richard Henderson (British Medical Research Council’s Laboratory of Molecular Biology in Cambridge, UK) were awarded the Nobel Prize in Chemistry for developing cryo-electron microscopy (Cryo-EM) for the high resolution structure determination of biomolecules in solution.
- Details
- Tuesday, 12 September 2017

Scientists who make movies of molecules in motion have a new high-speed camera to shoot with. The €1.2-billion (US$1.4-billion) European X-ray Free Electron Laser (XFEL) will start running its first experiments in September near Hamburg, Germany. The European XFEL fires powerful X-rays in bursts of a few hundred femtoseconds: so short that, like strobe lights, they can capture snapshots of jittery molecules frozen in time, and with a wavelength small enough to provide pictures at atomic resolution.
More Articles...
- Structural enzymology using X-ray free electron lasers
- Conformational landscape of a virus by single-particle X-ray scattering
- UWM researchers create first 3D movie of virus in action
- A Bright Future for Serial Femtosecond Crystallography with XFELs
- Experimental strategies for imaging bioparticles with femtosecond hard X-ray pulses





